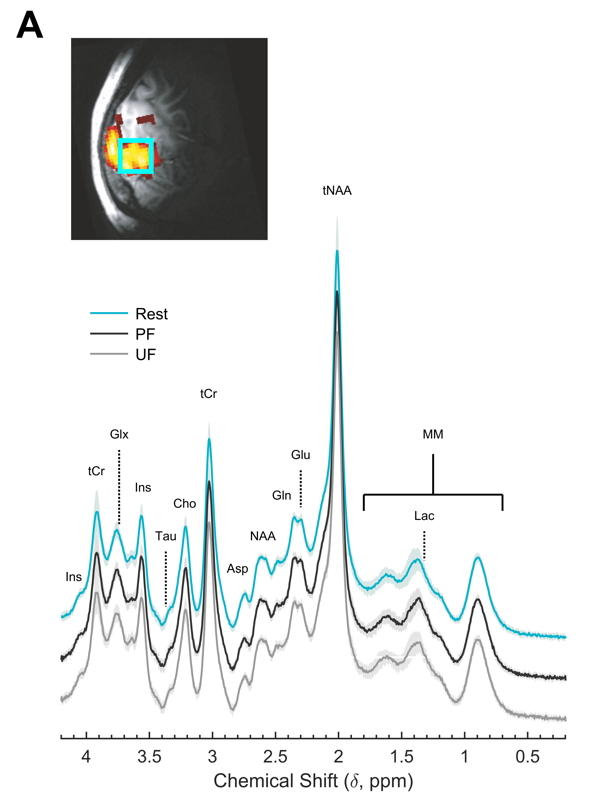
 Processing of incoming sensory stimulation triggers an increase of cerebral perfusion and blood oxygenation (neurovascular response) as well as an alteration of the metabolic neurochemical profile (neurometabolic response). Here we show in human primary visual cortex (V1) that perceived and unperceived isoluminant chromatic flickering stimuli designed to have similar neurovascular responses as measured by blood oxygenation level dependent functional MRI (BOLD-fMRI) have markedly different neurometabolic responses as measured by functional MRS. In particular, a significant regional buildup of lactate, an index of aerobic glycolysis, and glutamate, an index of malate-aspartate shuttle, occurred in V1 only when the flickering was perceived, without any relation with behavioral or physiological variables. Whereas the BOLD-fMRI signal in V1, a proxy for input to V1, was insensitive to flickering perception by design, the BOLD-fMRI signal in secondary visual areas was larger during perceived than unperceived flickering, indicating increased output from V1. These results demonstrate that the upregulation of energy metabolism induced by visual stimulation depends on the type of information processing taking place in V1, and that 1H-fMRS provides unique information about local input/output balance that is not measured by BOLD fMRI.
Processing of incoming sensory stimulation triggers an increase of cerebral perfusion and blood oxygenation (neurovascular response) as well as an alteration of the metabolic neurochemical profile (neurometabolic response). Here we show in human primary visual cortex (V1) that perceived and unperceived isoluminant chromatic flickering stimuli designed to have similar neurovascular responses as measured by blood oxygenation level dependent functional MRI (BOLD-fMRI) have markedly different neurometabolic responses as measured by functional MRS. In particular, a significant regional buildup of lactate, an index of aerobic glycolysis, and glutamate, an index of malate-aspartate shuttle, occurred in V1 only when the flickering was perceived, without any relation with behavioral or physiological variables. Whereas the BOLD-fMRI signal in V1, a proxy for input to V1, was insensitive to flickering perception by design, the BOLD-fMRI signal in secondary visual areas was larger during perceived than unperceived flickering, indicating increased output from V1. These results demonstrate that the upregulation of energy metabolism induced by visual stimulation depends on the type of information processing taking place in V1, and that 1H-fMRS provides unique information about local input/output balance that is not measured by BOLD fMRI.
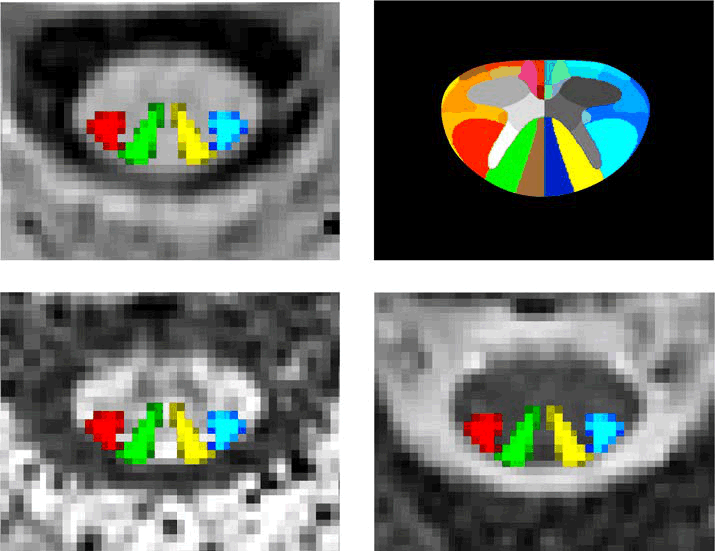 Quantitative spinal cord (SC) magnetic resonance imaging (MRI) presents many challenges, including a lack of standardized imaging protocols. Here we present a prospectively harmonized quantitative MRI protocol, which we refer to as the spine generic protocol, for users of 3T MRI systems from the three main manufacturers: GE, Philips and Siemens. The protocol provides guidance for assessing SC macrostructural and microstructural integrity: T1-weighted and T2-weighted imaging for SC cross-sectional area computation, multi-echo gradient echo for gray matter cross-sectional area, and magnetization transfer and diffusion weighted imaging for assessing white matter microstructure. In a companion paper from the same authors, the spine generic protocol was used to acquire data across 42 centers in 260 healthy subjects. The key details of the spine generic protocol are also available in an open-access document that can be found at https://github.com/spine-generic/protocols . The protocol will serve as a starting point for researchers and clinicians implementing new SC imaging initiatives so that, in the future, inclusion of the SC in neuroimaging protocols will be more common. The protocol could be implemented by any trained MR technician or by a researcher/clinician familiar with MRI acquisition.
Quantitative spinal cord (SC) magnetic resonance imaging (MRI) presents many challenges, including a lack of standardized imaging protocols. Here we present a prospectively harmonized quantitative MRI protocol, which we refer to as the spine generic protocol, for users of 3T MRI systems from the three main manufacturers: GE, Philips and Siemens. The protocol provides guidance for assessing SC macrostructural and microstructural integrity: T1-weighted and T2-weighted imaging for SC cross-sectional area computation, multi-echo gradient echo for gray matter cross-sectional area, and magnetization transfer and diffusion weighted imaging for assessing white matter microstructure. In a companion paper from the same authors, the spine generic protocol was used to acquire data across 42 centers in 260 healthy subjects. The key details of the spine generic protocol are also available in an open-access document that can be found at https://github.com/spine-generic/protocols . The protocol will serve as a starting point for researchers and clinicians implementing new SC imaging initiatives so that, in the future, inclusion of the SC in neuroimaging protocols will be more common. The protocol could be implemented by any trained MR technician or by a researcher/clinician familiar with MRI acquisition.
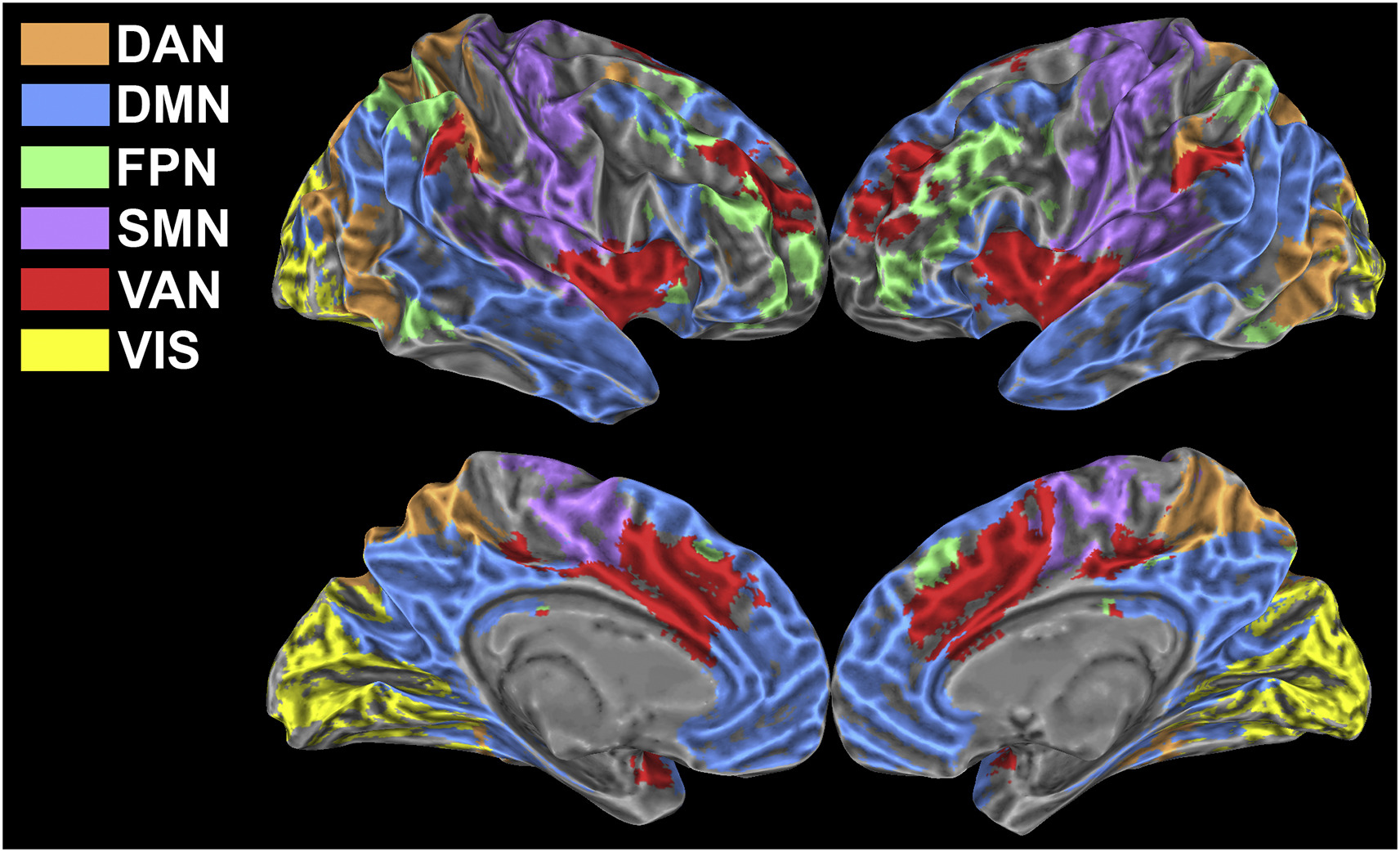 Brain activity at rest is characterized by widely distributed and spatially specific patterns of synchronized low-frequency blood-oxygenation level-dependent (BOLD) fluctuations, which correspond to physiologically relevant brain networks. This network behaviour is known to persist also during task execution, yet the details underlying task-associated modulations of within- and between-network connectivity are largely unknown.
Brain activity at rest is characterized by widely distributed and spatially specific patterns of synchronized low-frequency blood-oxygenation level-dependent (BOLD) fluctuations, which correspond to physiologically relevant brain networks. This network behaviour is known to persist also during task execution, yet the details underlying task-associated modulations of within- and between-network connectivity are largely unknown.
In this study we exploited a multi-parametric and multi-scale approach to investigate how low-frequency fluctuations adapt to a sustained n-back working memory task. We found that the transition from the resting state to the task state involves a behaviourally relevant and scale-invariant modulation of synchronization patterns within both task-positive and default mode networks. Specifically, decreases of connectivity within networks are accompanied by increases of connectivity between networks. In spite of large and widespread changes of connectivity strength, the overall topology of brain networks is remarkably preserved. We show that these findings are strongly influenced by connectivity at rest, suggesting that the absolute change of connectivity (i.e., disregarding the baseline) may not be the most suitable metric to study dynamic modulations of functional connectivity. Our results indicate that a task can evoke scale-invariant, distributed changes of BOLD fluctuations, further confirming that low frequency BOLD oscillations show a specialized response and are tightly bound to task-evoked activation.
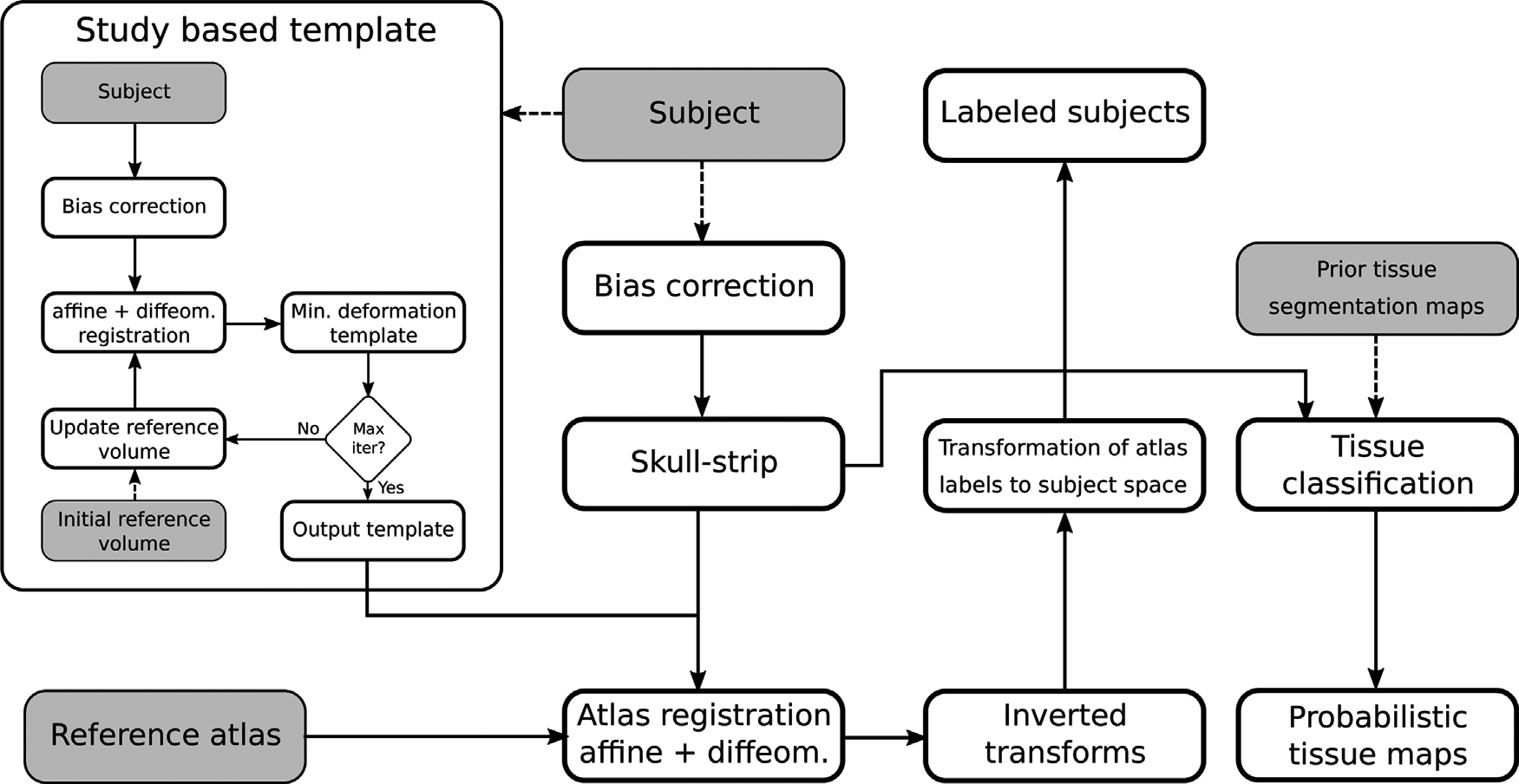 One of the most common tasks in small rodents MRI pipelines is the voxel-wise segmentation of the volume in multiple classes. While many segmentation schemes have been developed for the human brain, fewer are available for rodent MRI, often by adaptation from human neuroimaging. Common methods include atlas-based and clustering schemes. The former labels the target volume by registering one or more pre-labeled atlases using a deformable registration method, in which case the result depends on the quality of the reference volumes, the registration algorithm and the label fusion approach, if more than one atlas is employed. The latter is based on an expectation maximization procedure to maximize the variance between voxel categories, and is often combined with Markov Random Fields and the atlas based approach to include spatial information, priors, and improve the classification accuracy. Our primary goal is to critically review the state of the art of rat and mouse segmentation of neuro MRI volumes and compare the available literature on popular, readily and freely available MRI toolsets, including SPM, FSL and ANTs, when applied to this task in the context of common pre-processing steps. Furthermore, we will briefly address the emerging Deep Learning methods for the segmentation of medical imaging, and the perspectives for applications to small rodents.
One of the most common tasks in small rodents MRI pipelines is the voxel-wise segmentation of the volume in multiple classes. While many segmentation schemes have been developed for the human brain, fewer are available for rodent MRI, often by adaptation from human neuroimaging. Common methods include atlas-based and clustering schemes. The former labels the target volume by registering one or more pre-labeled atlases using a deformable registration method, in which case the result depends on the quality of the reference volumes, the registration algorithm and the label fusion approach, if more than one atlas is employed. The latter is based on an expectation maximization procedure to maximize the variance between voxel categories, and is often combined with Markov Random Fields and the atlas based approach to include spatial information, priors, and improve the classification accuracy. Our primary goal is to critically review the state of the art of rat and mouse segmentation of neuro MRI volumes and compare the available literature on popular, readily and freely available MRI toolsets, including SPM, FSL and ANTs, when applied to this task in the context of common pre-processing steps. Furthermore, we will briefly address the emerging Deep Learning methods for the segmentation of medical imaging, and the perspectives for applications to small rodents.
(c) 2016-2018 Centro Fermi